By Simon Brendel (ERNI Germany)
In medical device development, usability engineering often plays a minor role, treated as a downstream task before regulatory submission. Yet integrating usability early creates safe products that stand out. This article shows how usability drives efficiency, quality and market success, even in regulated environments.
Usability – Often underestimated, rarely done right
Regulatory requirements – such as those defined in IEC 62366, MDR (Medical Device Regulation), IVDR (In Vitro Diagnostic Medical devices and Repealing Directive) or the FDA Human Factors Guidance – demand a minimum level of usability, primarily aimed at avoiding harm. However, meeting these requirements alone is not enough to create a good product. These standards require that risks are addressed – but not that a product is intuitive, efficient or even enjoyable to use. This underestimation often leads to usability activities being started too late or approached half-heartedly in the MedTech context – resulting in costly redesigns or, in the worst case, flawed products.
Start early, prioritise wisely
Whether it’s chief physicians, lab workers or nurses – every system has multiple user groups with different tasks and needs. Ignoring this diversity puts the acceptance of the product by entire user groups at risk – and can ultimately lead to failure.
Another major barrier to establishing genuine usability across the MedTech sector is that the people who actually use the products are often not the ones who decide which product to buy. Instead, purchasing decisions are typically made by procurement teams or clinical leadership – individuals who rarely work directly with the devices. As a result, usability rarely becomes a decisive factor in purchasing decisions, even though it plays a critical role in daily clinical routines when it comes to safety, efficiency and acceptance. In fact, efficiency gains achieved through good usability can make a significant economic difference in the long term. So why isn’t this topic given the attention it deserves?
Compliance as a byproduct of good processes
Usability engineering should be an integral part of a solid requirements process – not something treated as a nice-to-have. Teams that analyse user contexts early, develop task models, create personas, and test iteratively will naturally generate the artifacts needed for regulatory approval. These deliverables don’t arise from a sense of obligation, but as a logical outcome of a sound development process – grounded in real insights, not assumptions.
From users to artifacts: A lean process
Figure: The ERNI Medical Usability Process builds on the human-centred design process defined in ISO 9241-210 and adds key elements required for the approval of medical devices.
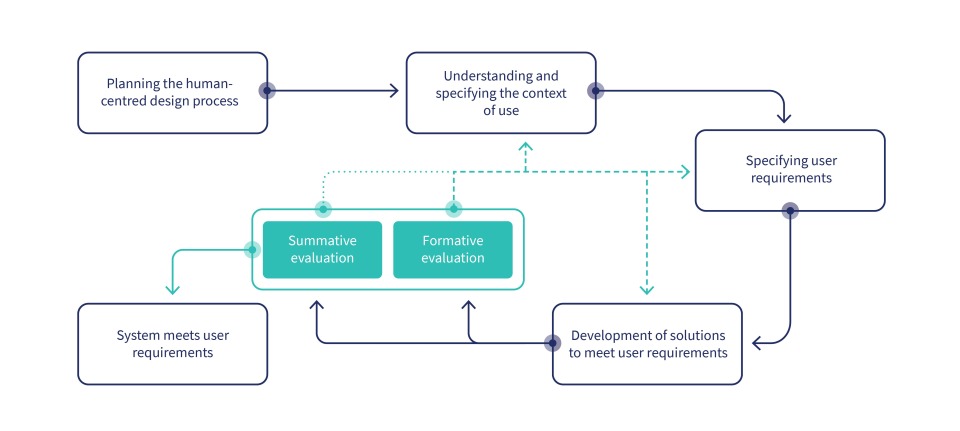
The process we developed to prevent real usability engineering from being neglected in MedTech projects is deliberately based on the human-centred design process as defined in ISO 9241-210. This approach is widely used in industries where high-quality usability has historically been given far more weight than in the medical device field, such as e-commerce, gaming and entertainment or B2C app development.
Our process follows the classic steps of human-centred design: planning, understanding and specifying the context of use, specifying the user requirements, and producing design solutions. When evaluating prototypes and other development artifacts, we differentiate between formative and summative evaluations. Our approach works equally well within V-models, agile environments and hybrid frameworks.
If all steps are executed carefully and at the right time, the process naturally results in both widely accepted usability deliverables and all artifacts required by regulatory standards:

Usability engineering is requirements engineering
In practice, usability engineering and requirements engineering are difficult to separate. Insights gained through human-centred design – interviews, observations and context analyses – directly feed into requirements. Many findings from usability activities become critical inputs for defining requirements. That’s why these two disciplines should be closely integrated, ideally combined in a single role or a tightly aligned team. A project without a dedicated usability lead misses out on valuable potential – and risks failure.
In order to align both disciplines effectively and to ensure good product design is not lost in the face of perceived regulatory complexity, it is important to regularly reflect on the following questions:

Conclusion
Usability is not an add-on for regulatory approval; it is a strategic success factor, provided it is taken seriously. In our projects, we often see the differences between formally fulfilled requirements and true usability. With the ERNI Medical Usability Process, we support our clients in asking the right questions from the very beginning, consistently involving users and viewing usability not as a duty, but as a tool. That’s how we jointly create products that are not only safe but also compelling and commercially successful.


